 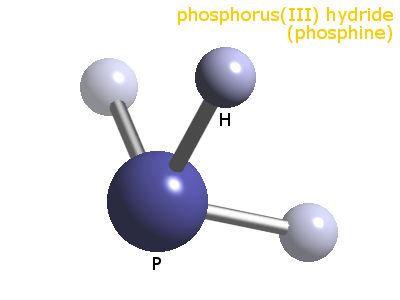 
In contrast, ammonia shows trigonal pyramidal geometry and <109 bond angle, and the reason behind this deviation is explained by the VSEPR theory. The expected bond angle is 109.28 degrees, and the geometry is tetrahedral. If the number of hybridisation is four, then the hybridisation of the central atom is sp3. In ammonia, the sum of the lone pair and the bond pair is 1+3 = 4 N in NH3 has three bonds around it and a lone pair.Īccording to the VSEPR theory, the sum of the number of lone pairs and bond pairs of the central atom gives the hybridisation of the molecule. Valence shell electron pair repulsion theory states that the shape and geometry of a molecule are decided by the repulsions existing between the bond pair electrons and lone pair electrons of the central atom. This cannot be explained by the VBT, but can be explained by the VSEPR theory. The actual geometry for ammonia is trigonal pyramidal, and its bond angle is 107 degrees. Out of the four sigma bonds, three are with an unpaired electron, and one is with lone pair electrons.Īccording to VBT, for the sp3 hybridisation, geometry is tetrahedral, and the bond angle is 109.28 degrees. It uses 2s and 2p orbitals in the valence shell and undergoes sp3 hybridisation to give four hybrid orbitals and overlap head-on and lead to sigma bonds. In NH3, N has 1s22s22p3 electronic configuration. The valence bond theory explains the bonding in the molecule with the overlapping of orbitals in the valence shell. GeometryĪmmonia molecule geometry is explained by valence bond theory (VBT) and the valence shell electron pair repulsion (VSEPR) theory. Ammonia and compounds of ammonia can show their basic nature turning red litmus blue. Ammonia can exist as liquid as well as gas. Ammonia is a base that has a pungent smell. Nitrogen is the central atom in Ammonia and H are the bonded atoms around nitrogen. NH3 is the molecular formula for ammonia. The number of electrons that can be shared between two atoms can be 2,4 or 6, where 2 is minimum, and 6 is maximum. The electrons that do not participate in the bonding but are present in the valence shell of the central atom is called a lone pair. The coordinate covalent bond is a bond similar to a covalent bond, but one of the atoms in the molecule should have an extra pair of electrons like lone pair. This type of sharing is called coordinate covalent bondingĬovalent bonding can be seen between two atoms with equal electronegativity or with less difference in electronegativity or two non-metals. The two electrons are shared between the two atoms. The sharing of electrons in which one of the bonding atoms contributes two electrons, and the other atom contributes no electrons.The first type is where the bonding atoms contribute one electron each and share the two electrons among the atoms, known as covalent bonding.The sharing of electrons can be of two types between the atoms: Elements present in these compounds share electrons in various ways and form bonds between them. Molecules that contain different atoms in them are called compounds. Molecules formed between different elements can be covalent or ionic.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
