A107 4877Ĭhattaraj P K and Sarkar U 2003 Chem. 391 138 (for recent work on molecules)Ĭhattaraj P K and Sarkar U 2003 J. Sako T, Cer-nusak I and Diercksen G H F 2004 J. 70 417, and references therein for confined atom calculations 105 5839ĭolmatov V K, Baltenkov A S, Connerade J P and Manson S T 2004 Radiat. 64 701įröman P O, Yngve S and Fröman N J 1987 Math. Kouwenhoven L P, Austing D G and Tarucha S 2001 Rep. 19 137īányi L and Koch S W 1996 Semiconductor quantum dots (Singapore: World Scientific) Xiao Z, Zhu J and He F 1996 Superlattices Microstruct. Laux S E, Kumar A and Fischetti M V 2004 J. Zicovich-Wilson C, Planelles J and Jaskólski W 1994 Int. B31 3557Ĭonnerade J P, Dolmatov V K and Lakshmi P A 2000 J. 325 29Ĭonnerade J P and Dolmatov V K 1998 J. Sen K D, Garza J, Vargas R and Vela A 2000 Chem. 
90 3377īaderabd RFW and Austen MA 1997 J. 71 2235Īquino N, Flores-Riveros A and Rivas-Silva J F 2003 Phys. Ten Seldam C A and DeGroot S R 1952 Physica 18 891 54 107Ĭruz S, Ley-Koo E, Marin J L and Taylor-Armitage A 1995 Int. 74 4603įernández F M and Castro E A 1982 Kinam 4 193 32 56ĭe Groot S R and Ten Seldan C A 1946 Physica 12 669 Michels A, De Boer J and Bijl A 1937 Physica 4 981 Parr R G, Donelly R A, Levy M and Palke W E 1978 J. Parr R G and Yang W 1989 Density-functional theory of atoms and molecules (New York: Oxford University Press) 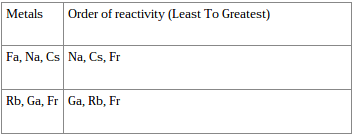
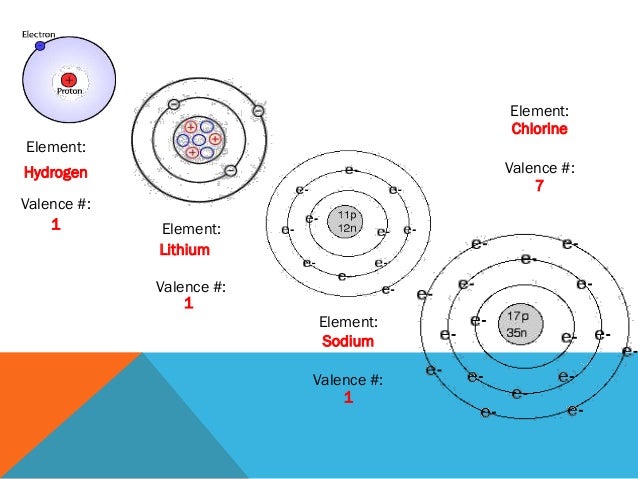
General trends of the atomic softness parameter under spherically confined conditions are reported and discussed. At a certain confinement radius, the atomic global hardness is even reduced due to such crossover. While the global hardness of a confined atom increases as the radius of confinement decreases, due to the accompanying orbital energy level crossing, it does not increase infinitely. The electronegativity in a confined atom is found to decrease as the radius of confinement is reduced suggesting that relative to the free state the atom loses its capacity to attract electrons under confined conditions. As the atoms move from left to right, the electronegativity of these atoms are decreasing as the distance between the electrons and the positively charged nucleus is would be greater, giving it lower ability to attract electrons. Here is a visual explanation of the situation. With this boundary condition the non-relativistic spin-polarized Kohn-Sham equations were solved. Whereas an elements reactivity is based more on how ready it is to gain or lose electrons. The atomic electrons are confined within the impenetrable spherical cavity defined by a given radius of confinement satisfying the Dirichlet boundary condition such that the electron density vanishes at the radius of confinement. The density functional descriptors of chemical reactivity given by electronegativity, global hardness and softness are reported for a representative set of spherically confined atoms of IA, IIA, VA and VIIIA series in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
